The impact suspended solids pose on chillers
We need to recognise that the contamination of closed systems can never be fully prevented.
The actual history of a system can often be very difficult to determine, with legacy contaminates often poorly reported. Further introduction of organic and inorganic ‘dirt’ (contamination) adds to this collective over time. Even newly commissioned systems, if not meticulously monitored, are often seen with corrosion debris due to ‘flash’ corrosion during filling.
The chiller installation process itself can introduce all manner of dirt, dust, filings, oils, grease, swarf, flux residues as well as potential bacterial issues, especially if systems have been left untreated and allowed to stagnate for any length of time between commissioning and operation. Equally over time, more established systems can become fouled with scale (hard water ingress), corrosion (from poorly treated systems) and again from increased microbiological activity.
Fig 1. Heavily fouled with solids

To help assess the levels of ‘dirt’ in the form of solids in our closed water system we can test for what is referred to as suspended solids. In general, suspended solids are particles present in water but for our specific purposes, we will understand them as solid particles that remain suspended in water and have a particle size of 2 microns or larger.
You might also hear the phrase ‘dissolved solids’, which means certain particulates smaller than our 2-micro differentiator, and opposed to what we are discussing in this bulletin. For background information, dissolved solids do have a role to play when considering the conductivity of water within a closed circuit; as totally dissolved solids (commonly referred to as TDS) they are readily used to monitor variations within a system such as concentration of water treatment inhibitors, glycol, and the like.
If the presence of suspended solids is elevated beyond the maximum guidance threshold of >30 mg/litre and excessive suspended solids are not removed, they can have a multitude of adverse effects on a chilled circulating system that include but are not limited to:
- Energy efficiency that directly increases running costs
- Blockages leading to ‘dead spots’ around the system.
- System erosion (generating further suspended solids, adding to the rate of erosion).
- Clogging by sludge debris of heater exchanger, which puts the exchanger at risk of localised failure.
- Increased wear on pumps that can lead to possible leaks and failure.
- Reducing the effectiveness of water treatment chemicals thus increasing corrosion potential.
- Encouraging bacteria proliferation, made possible by creating ideal growth conditions.
MONITORING MEASUREMENT AND CONTROL
Addressing the presence of suspended solids becomes a key part of any system management strategy. The *BSRIA BG 50/2021 standard and guidance states that suspended solids should be controlled at less than 30 mg/litre, that ‘in the circulating water and a well-controlled system consistently achieve less than 10mg/litre’. As much as ‘low’ suspended solid values are a good measure of system integrity, we need to be mindful that it does not guarantee that active corrosion is not occurring from elsewhere in the system, hence strategic sampling needs to be considered, as shown in Table B.
In consideration of individual reported levels of solids, any trends upward in solids over time also needs to be monitored as this is likely a direct result of internally corroded metals from the system. It is worth mentioning that as much as periodic water testing provides information on changes to water quality, it is limited. This alone does not provide evidence of the actual condition of the physical system. Providing this additional system evidence can be achieved by utilising equipment such as **corrosion coupon racks and/or online electronic sensors. These devices are extremely useful, acting as an early warning of active corrosion, which provides actual rates of system losses. Strategies expressed here are recognised within guidance as part of an overall means of monitoring and control.
It is accepted that low level rates of corrosion are very difficult to avoid. This is recognised by the guidance that offers predefined millimetre-per-year thresholds against various metals typically found in such systems – see Table A. In a well-controlled system, however, corrosion rates should be well below these ‘threshold’ values and shouldn’t necessarily be seen as maximum control limits.
TABLE A.

A simple indicator of increased levels of suspended solids is often seen as a ‘cloudy’ sample of water, as a colouration of a collected water sample. This is why a visual evaluation is included in the ‘analysis’ undertaken during routine water treatment sampling.
The insoluble nature of these solids means that we can readily measure the levels of total suspended solids (TSS) within a system using a solids recovery filtration method (gravimetric). This method of measurement is seen as more accurate and preferred by laboratories.
Fig 3: Example of corrosion rack housing the coupons

There are other, quicker and less accurate means of TSS measurement; colourimetric for example, which utilises an electronic, small, handheld device and the transmission of light to determine the amount of TSS, yielding an immediate result. One further minor advantage of using colourimetric devices is that they can detect at a lower (20mg/l) level.
This type of device certainly offers a quick initial indicative result, however, this method is suspectable to interference, caused for example by entrapped air bubbles that can interfere with the final result. Certainly, if this method of measurement is being considered as a way of routine testing, then it is worth performing against the laboratory-based gravimetric analysis using a number of individual tests to check whether they correlate, offering a means of interpretation.
As referenced above, the gravimetric and preferred (more accurate) means of TSS measurement in water is a method of analysis which is described by the ***Blue Book. In simple terms, a fixed volume of water is filtered under a vacuum with those solids of a certain size retained on the filter medium. The residue material is dried in an oven, removing all moisture and then weighed. This final weight of remaining solids is reported as the TSS of that water sample.
As much as this process will give us an indication of TSS in a system, the larger and more complex the systems are, the higher the potential for variability of TSS. These systems are dynamic, and as such subject to velocity variability, which has a bearing on our results. Certainly, gravity, coupled with a poor flow profile for larger particulates may result in settlement and pockets of concentration of solids locally.
In recognition of volume size, and increased variability, guidance (**BG 50) recommends an increase in sample locations for a more holistic overview. Below is an extracted table referenced in BG 50 guidelines, which lists how increasing volumes are met by a wider sampling scope:
TABLE B.
It is important to be able to recognise that should samples be taken from areas of low velocity (for example terminal units or the pipework feeding them), then these samples may contain ‘settled’ solids and as such would contribute to reports of elevated total suspended solids. Unfortunately, laboratory testing would not be able to differentiate the difference between ‘suspended’ and ‘settled’.
In situations where higher overall counts are recorded, localised flushing may be recommended to remove the excessive build-up of such ‘settled’ solids. If localised flushing were to be undertaken, regular testing of these points would be also recommended. If the build-up of ‘settled’ solids reappeared after two—three weeks, for example, then this it would indicate a wider potential risk of active corrosion occurring and further system wide investigation would be warranted.
SUSPENDED SOLIDS REMOVAL
Fortunately, should our systems show signs of increased suspended solids, corrosion, or other debris, there are a number of devices that can be used to remove such contamination. This includes strainers, filters, magnetic filters, dirt separators, combined air and dirt separators as well so commonly found side stream filtration. All these have a role to play, and when applied correctly can have a positive impact on cleaning and continued protection of closed circuits.
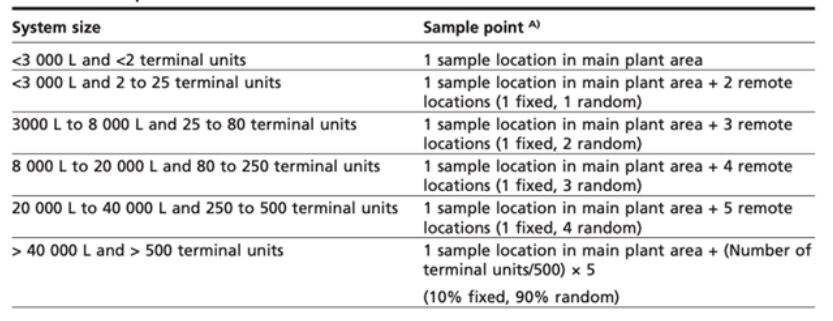
A number of systems have side-stream filtration included at the design stage in order to safeguard some of the issues we have highlighted above. Such equipment is simple easy to install and has a simple process of solids removal. Side stream filters are designed to constantly pass 5-15% of the circulating water through a filter housing (see diagram) resulting in the total system volume being filtered 2 to 3 times every 24 hours.
Differing grades are available and where a system has known issues, a courser filter is typically chosen to remove the larger particles.
Once the courser filter has removed these larger particles, finer filters are applied systematically. This stops the ‘blinding’ of finer filters and allows for a more controlled-step approach to debris removal. Getting down to a filter size of 30 microns offers good results.
Where a system has experienced high levels of contamination, it is good practice to utilise specific water treatment chemicals to aid the recovery. Dependent on the issue(s), a series of
chemical products can be selected. We can help evaluate the correct procedure and select the chemicals to best address such issues.
For example, scaling would require using an acid-based cleaning agent, microbiological issues are treated with selected biocides, and corrosion using a dispersant. Depending on the severity of the contamination, full chemical flushing can be avoided as these can be potentially very disruptive and costly.
Prevention is always better than cure. Where lower levels of debris are recorded, it is possible to undertake an online clean, as described above, with a mix of chemical treatment and filtration. As part of the metal surface cleaning process, it is critical to passivate the newly cleaned surface by the creation of a protective layer of magnetite. This is followed by the addition of corrosion inhibitors that disrupt the process of corrosion, making the surface corrosion resistant.
Once again, our engineers can help with the selection and application of these protective products.
OTHER CLEANING SYSTEMS
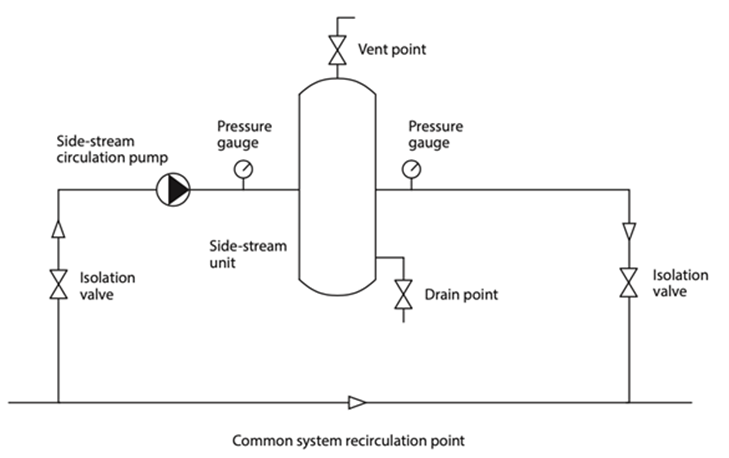
Strainers
These are simple coarse solid housed filters mainly used to collect larger suspended matter, used primarily to protect pumps and control valves. They are installed inline, in front of the equipment or devices they are to protect etc. They should be subject to regular inspection and cleaning.
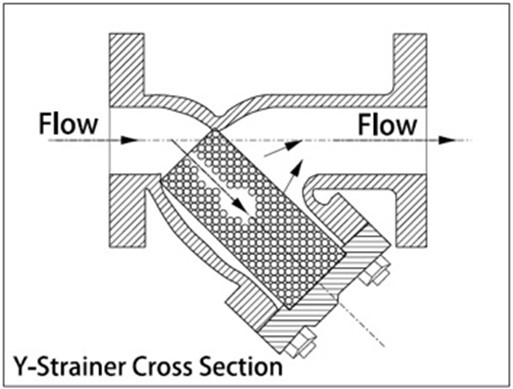
Dirt separators
This approach to ‘catch’ the debris is, again, a simple theory, involving slowing down the velocity of circulating water, coupled with allowing the flow of water and any suspended debris to impact on a mesh grid housed in an in-line vessel. This twinned process encourages the settling out of larger particulates, which ‘fall’ to the base of the separator unit – hence the term ‘dirtseparator’. Routinely these units are drained from the base to clear out any settled (caught) particles.

Magnetic separators
If the corrosion has built up in the circulating water, the inclusion of a magnet to attract any suspended corroded matter is a highly efficient method of capture and removal. These separators house extremely powerful magnets which attract metallic material regardless of size. This is important, as, for example, magnetite (iron oxide) has an average particle size of 8 μm, which is substantially lower than the physical filter sizes that can be practically used in chiller systems. There are filter systems that additionally house such magnets within the flow pathway so that each pass has a maximum efficiency in solids removal.
Please contact us for advice and support on water treatment to maximise your systems’ performance, reduce overall running costs, and help extend it’s lifecycle.
Author: Simon Mattock, Advanced Water Technologies (AWT).
Related Articles

March 11, 2026
TCUs Don’t Fail. Water Kills Them
Why water treatment is the most overlooked service in plastics moulding cooling and temperature control — and what it costs when it’s ignored Here is...
Read More
May 15, 2025
The Hidden Costs of Poor Water Treatment in Cooling Systems
Efficiency, reliability, and longevity are critical considerations in managing closed-circuit process chillers. While these units form the backbone of commercial and industrial cooling systems, their...
Read More
May 6, 2025
Preparing Your Chiller for Summer
Summer can be an unreliable season, often bringing unexpected heatwaves. Just as an overheated car engine can cause significant issues, the loss of cooling capability...
Read More