As much as we need protection from cold weather and the effects it can have on us, our homes, our cars etc, we need to protect our chillers. It can be very costly if pipework splits due to the lack of frost protection. If not appropriately addressed beforehand, once temperatures drop below zero, we can quickly get into a situation which is uncoverable.
In this paper, we mainly focus on the use of chemicals to protect chiller systems from freezing, but it should be mentioned that trace heating is also a means of protecting pipes from freezing.
When considering chemical protection, one may instinctively think of commonly, over-the-counter anti-freeze that can be used in cars, and they would be right. However, commonly used automotive anti-freeze is not designed or formulated for the types of complex mixed metal systems that we find in industrial applications.
In addition, such products contain additives such as silicate that are incompatible with chiller systems. Indeed, silicates readily coat metal surfaces and with that reduce the efficiency of heat transfer (typically + by around 10%), which adds to overall energy costs.
Silicate, in the right environment, can also form polymers, turning fluid into a gel, again adding further economic cost and possible fouling. The final warning not to use such products is the abrasive nature of silicates, which can eventually attack pump seals.
What we should avoid, what products can we select from and in what concentrations?
Generally, there are two commercially available products designed for use to protect industrial closed water systems from freezing.
Both are formulations that contain glycol:
- Mono-Ethylene Glycol (commonly referred to as MEG) is the most popular. MEG is seen as the commonly used antifreeze for industrial applications where there is no special requirement or need for a low-toxicity chemical. The main benefit is that due to its consistency, it has the best heat transfer profile rate and with that helps reduce ongoing energy costs.
- Mono-propylene Glycol (commonly referred to as MPG). MPG may not have the level of heat transfer efficiency of the MEG, but is considered suitable for use in food factories/pharmaceutical manufacturers, being recognised as a ‘food grade’ chemical product. As MPG is less efficient than MEG, therefore often requires a ‘correction’ to allow for this differential that affects cooling capacity. This is typically managed by reducing the chiller settings and with that increasing plant workload and thus eventually increasing energy costs.
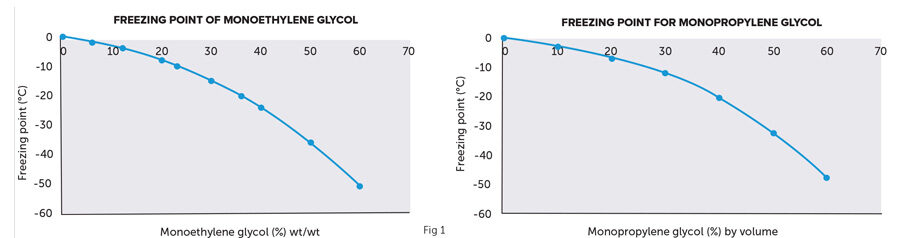
The aforementioned types of glycol are recommended as part of any commissioning process and/or by the relevant chiller manufacturer. The concentration of glycol will be determined by the level of freezing-point protection required. Typical protection levels for closed systems in the UK require minimum glycol concentration levels of 25% v/v, which would provide a freeze protection average of -12°C. Fig 1 graphs express the concentration vs freezing point protection correlation for both MEG and MPG.
The additional consideration for achieving a minimum concentration of 25% relates directly to the suppression of bacteria. Glycols are carbon-based, which means as a food source they are susceptible to attack from bacteria. However, at levels of 25% and beyond, such concentration creates what is known as a biostatic status and as such inhibits the proliferation of microorganisms associated with the breakdown of glycol (fig2).
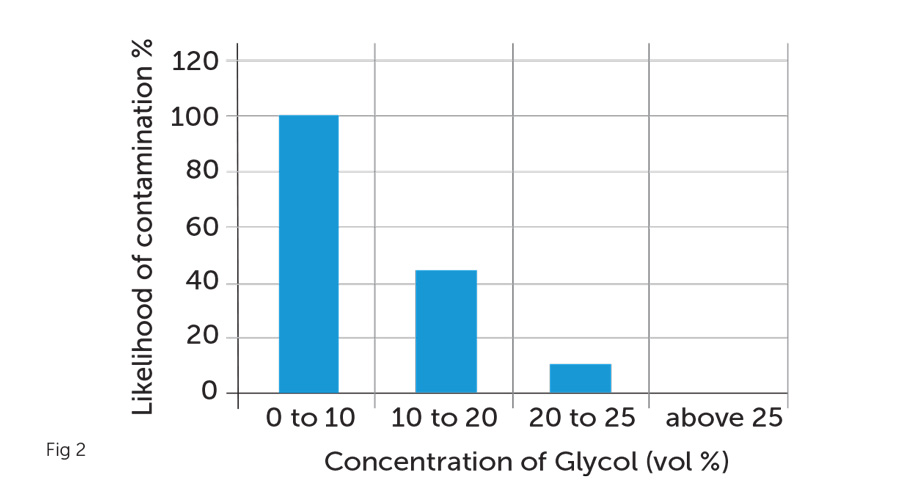
Should such a breakdown occur due to their presence and growth within the water system, not only will we have lost the correct levels of anti-freeze protection, but this bacteriological activity will also have the effect of driving down system pH levels, thus leading to increased corrosion. This manifestation is due to microbiological by-products which are created in the food-to energy conversion chain.
On the other hand, ‘overdosing’ of glycol brings with it its own challenges. Too much glycol will see efficiency levels drop due to the increase in the power needed to circulate the fluid contained within the system, as well as the reduction in heat transfer performance, again adding to an increase in overall running costs.
What other considerations should we think about when using a chemical antifreeze?
If we get back to basic principles, we might need to avoid using direct mains-fed water. This is due to the varying quality we see across the UK, which includes the concentration of deposits and additives that, given the right conditions, can damage chilled pipework.
To help combat certain quality issues we might need to consider filtering and adding a non-oxidising biocide to our water source. In addition to pre-conditioning of the water being used, we will need to add appropriate additional chemicals that will help protect chillers and their multi-metal components. These types of chemicals are typically referred to as inhibitors. Both water and glycol have properties, which without adding chemical inhibitors, can promote corrosion, and depending on the area of the country, the scaling of pipework if the water used is ‘hard water’*.
These properties can all contribute to serious issues over time. Inhibitors are carefully selected depending on the metals used in a system. They become a vital component in the efficient running of a chiller, which will also help extend the system’s lifecycle. Such inhibitors can be included in a mix of what is referred to as inhibited glycol, which is a common approach seen across the UK. These two chemical components can also be added separately as the situation dictates, with tailored levels of both glycol and inhibitor administered.
Preventative maintenance sampling
Once we have added our glycol and inhibitor (and if needed nonoxidising biocide) we should not overlook the need for regular maintenance sampling. Once an investment has been made to achieve the optimum blend of glycol and inhibitor to protect our system, we need to closely monitor that they remain intact, retaining the right concentrations.
We also learn from maintenance sampling how stable are the conditions within the system, whether we are seeing an increase in bacterial activity, or any active corrosion, pH depression etc.
This maintenance sampling regime is key to avoiding costly system failures and keeping the entire system as energy efficient as possible.
As part of our planned preventative maintenance contracts, ICS Cool Energy now carry out as standard, water sampling and independent laboratory analysis. We take a sample, send it off to an independent laboratory and provide you with an easy to-read report indicating any defects which fall outside of the recommended parameters.
There is also clear advice to maintain your equipment or carry out a repair, all of which we do in house, be it glycol, inhibitor, biocide or a full flush.
Related Articles

February 15, 2024
5 Tips To Reduce Your Process Cooling Energy Costs
Refrigeration accounts for a high proportion of overall site energy costs, most notably cooling for food, drink, chemical, and pharmaceutical processes where up to 90%...
Read More
November 29, 2023
ICS Cool Energy Enhances Preventive Maintenance Plans for Temperature Control Process and Industrial Equipment
New service packages help customers ensure their temperature control installations remain operational and efficient over their maximum lifespan. ICS Cool Energy, an international market leader...
Read More
July 7, 2023
How To Manage Suspended Solids
The impact suspended solids pose on chillers We need to recognise that the contamination of closed systems can never be fully prevented. The actual history...
Read More